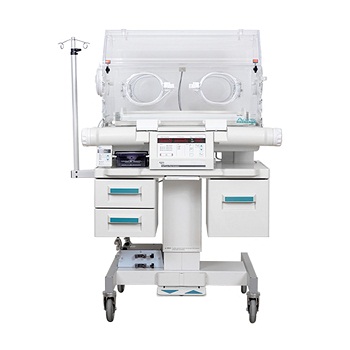
Baby Incubator Ohmeda USA
Baby Incubator Ohmeda USA
Baby Incubator Ohmeda USA is the exclusive product in Pakistan, New Baby Incubator Price is very high in Pakistan but GE Care Plus 4000 is an advanced incubator that provides superior thermo-regulation with an integrated micro processor based controller. Optional active double-wall construction creates a precise and consistent environment for the neonate. There are many Baby Incubators in Pakistan but Baby Incubator Ohmeda USA is the most used incubators in the world. For more information regarding the Baby Incubators in Pakistan and Baby Incubators Price in Pakistan, please feel free to contact us. CLICK HERE FOR BABY WARMERSFeatures:
- Microprocessor-based control system for accurate thermoregulation
- Self-test functions at power-up and during normal operation
- Patient control (Servo) mode and air control (Manual) modes
- Continuous positioning from 0° to 12°
- Optional active double-wall construction
Specification:
Dimensions:- Incubator with Elevating Base
- Height: 54 - 62 in (136.2 - 157.5 cm)
- Elevating base: 35 - 43 in (88.9 - 109.2 cm) mattress to floor
- Depth: 28 in (71.1 cm)
- Width: 33 in (83.8 cm)
- Weight: 195 lbs (89 kg)
- Incubator with Stationary Pedestal
- Height: 53 in (135.9 cm)
- Depth: 28 in (71.1 cm)
- Width: 33 in (83.8 cm)
- Weight: 180 lbs (80 kg)
- Drawer Packages for Elevating Base or Stationary Pedestal
- Overall: 11 x 11.5 x 18.25 in (28 x 29 x 46 cm)
- Single deep drawer: 10 x 9 x 16.5 in (25 x 23 x 42 cm)
- Dual drawer: 4.5 x 9 x 16.5 in (11 x 23 x 42 cm)
- Incubator with Cabinet
- Height: 55 in (141 cm)
- Depth: 25.5 in (64.6 cm)
- Width: 35 in (88.9 cm)
- Weight: 185 lbs (84 kg)
- Incubator Characteristics
- Casters: 5 inch diameter, 2 locking, 2 non-locking
- Mattress: 13.7 x 25.6 in (34.8 x 65 cm)
- Mattress to Hood: 16 in (41 cm)
- Door Height: 11 in (28 cm)
- Positions: Continuous from 0° to 12°
- Port holes: 6
- Tubing ports: 6
System Controls
- Microprocessor-based control system: Self-test functions are performed at power-up and during normal operation
- Patient Control (Servo) Mode: 95° - 98.6°F (35° - 37°C)
- Air Control (Manual) Mode: 73.4° - 98.6°F (20° - 37°C)
- Up to 102.2°F (39°C) with control panel override switch
- Air Velocity: Average < 10 cm/sec with inner wall
- Temperature variability: Less than ± 0.2°F (0.1°C)
- Resolution: ± 0°F (0.1°C)
- Probe or interchangeability: ± 0.2°F (0.1°C)
- Average Oxygen Input Concentration Range: 5 L/min 25-45% | 10 L/min 35-65% | 15 L/min 45 to 70%
- Humidification: Standard — 10 to 75% dependent on nursery environment and incubator temperature setting | Servo — 40 to 85% regardless of nursery environment and incubator temperature setting
Bistos Fetal Doppler BT200L Korea
Bistos Fetal Doppler BT200L Korea
Bistos Fetal Doppler BT200L Korea is a brand named HI-bebe, this product is showing you what fetus is saying. HI-bebe Bistos is pocket size Fetal Doppler for obstetricians, midwives and pregnant women. It has : BT-200L Mono LCD, 2MHz. which enables the user to figure out the heart rate of the fetus and gives absolutely astonishing results. Hi-Bebe Fetal Doppler BT200L is manufactured in Korea. Please click here to download the product catalog BT-200L.pdf Features: -- BT-200L Mono LCD, 2MHz
- High-sensitivity Doppler probe (2 / 3MHz available)
- Compact and light pocket Doppler
- Ergonomic design
- Large display and clean sound
- Easy to use
- Long time continuous use (5hours)
- Variable display option : color OLED, mono LCD, sound only
- Waterproof probe
- FDA Approved
- LCD Heart Rate Display with signal quality indicator
- LCD Display now has a backlight
- Low Battery Indicator
- Heart Rate Range: 50-240 bpm
- Accuracy ± 2%
- Ultrasound Frequency 2MHz
- Ultrasound Intensity 10mW/cm2
- Speaker Output Power 1.2W
- Built-in Loudspeaker
- Audio output for Earphone
- Auto Shut Off 5 minutes
- Auto Shut Off on signal 1 minute
- Power 1.5V Battery x 2 (Type: AA)
- Sensitivity From 10 - 12 weeks
- Battery Life 360 minutes
- Safety Standards CE Certified, EN ISO 13485:2003 compliant
- Conforms to UK and EC Medical Safety Requirements (Medical Directive 93/94/EEC), CE 0470